Staff with highly specialized skills support research and development activities for regenerative medicine products!
Our company conducts raw material testing, in-process control testing, and quality testing of final products for regenerative medicine products under GMP (GCPT) regulations.
【Quality Testing Items】
■ Sterility Test (JP17 Membrane Filter Method, Direct Method)
■ Endotoxin Test (JP17 Colorimetric Method, Turbidimetric Method, Gelation Method)
■ Mycoplasma Detection Test (JP17 NAT Method using Real-time PCR)
■ Residual Testing for Antibiotics, Growth Factors, etc. (LC-MS/MS, ELISA, etc.)
■ Soft Agar Colony Formation Test, Cell Proliferation Characteristic Analysis (In Preparation)
■ Validation of Testing Methods related to the above tests
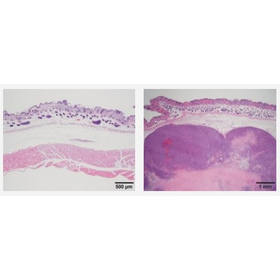
In a research facility dedicated to the safety evaluation of regenerative medicine products, we conduct general toxicity tests and tumorigenicity tests using immunodeficient mice.
【Non-Clinical Testing Items】
■ Tumorigenicity Test using Immunodeficient Mice
■ Cell Proliferation Characteristic Analysis, Soft Agar Colony Formation Test
■ General Toxicity Test
■ Safety Pharmacology Test, Efficacy Pharmacology Test
■ In Vivo Distribution Test (qPCR Method, Immunostaining)
■ Consulting for Regulatory Affairs and Applications is also available.